Corticosteroids cause osteoporosis and fractures in a high percentage of patients. There is a dose-dependent effect, which is difficult to define because of varying durations at each dose. The beneficial effects of steroids on the underlying disease may partially offset the detrimental effects on bone. Because the effects are so variable and can be clinically severe, patients embarking on long-term steroid treatment should have bone density monitored, and measures taken to try to preserve bone.

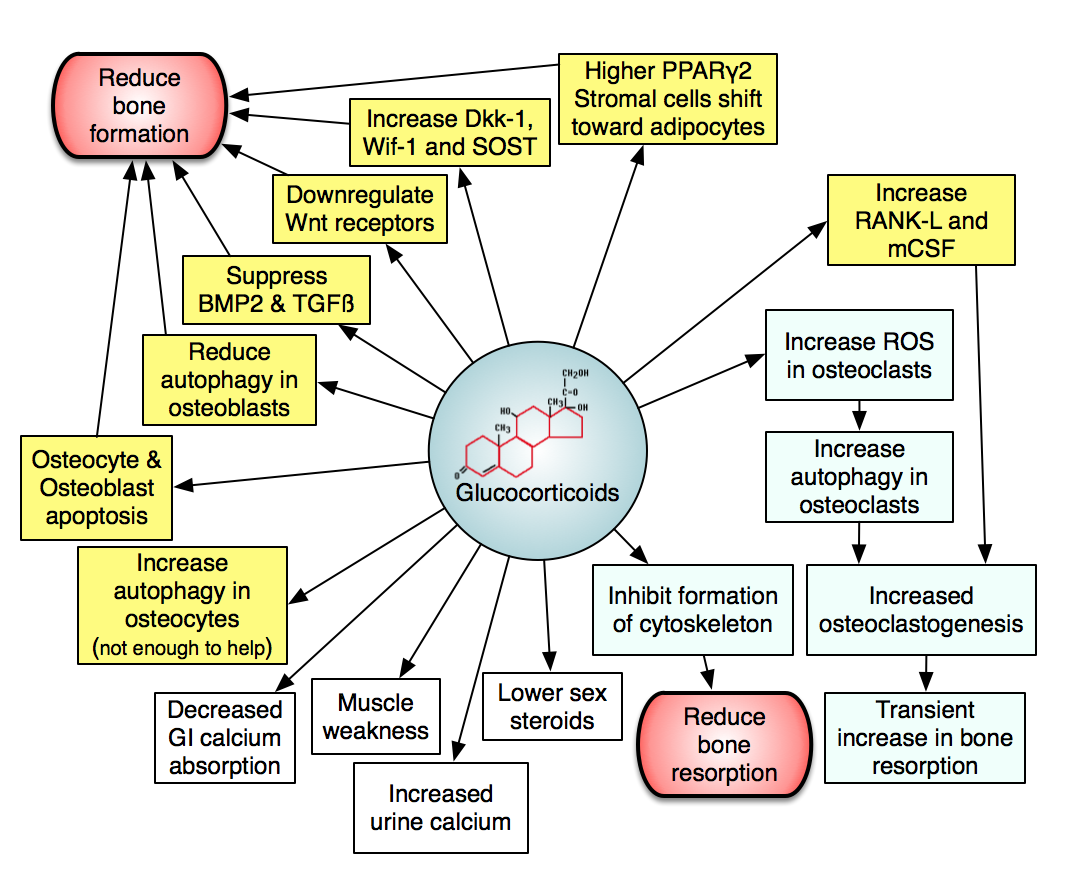
The figure shows effects of steroids on bone. The yellow boxes are effects on osteoblasts or osteocytes; the blue boxes are effects on osteoclasts, and the white boxes are other effects which will secondarily cause bone loss.
Here is a powerpoint presentation about this. I'm working on getting permission to use the figures that are cited.The trabecular bone is affected more rapidly than the cortical bone. Some patients suffer multiple vertebral compression fractures within a year of initiating steroid therapy.
Bone density should be measured in patients who are expected to remain on corticosteroids. The same lab tests that are used for "ordinary" osteoporosis are indicated for steroid-induced osteoporosis. In males it is very important to remember to check the testosterone level. Females should be asked about amenorrhea (which is probably a better indication of estrogen deficiency than estrogen levels). Measurement of LH or FSH will not be helpful in this situation, since the hypogonadism is usually hypogonadotropic, and thus low estrogen will not be associated with high FSH the way it is in a postmenopausal woman.
A 24-hour urine calcium is indicated, because often these patients have hypercalciuria. I usually check a urine N-telopeptide, too, to get an idea of how much of the hyercalciuria is due to bone resorption. Low urine calcium is also seen, especially if patients have any other reason for malabsorption. Vigorous treatment with calcium and vitamin D is too frequently undertaken without checking the urine calcium, and this could worsen hypercalciuria.
In patients with markedly low bone density, prevalent fractures, or high doses of steroids I usually also check a PTH and 25-hydroxyvitamin D level. In less severe cases I will just check these if the serum calcium or alkaline phosphatase is abnormal.
Xrays of the spine are especially important in patients taking long-term corticosteroids, because sometimes the fractures are not clinically obvious, and if they are present the patient will need maximal therapy.
The physiology of corticosteroid-induced osteoporosis is different than postmenopausal osteoporosis, so treatment cannot be expected to have the same results. For example, anti-resorptive therapy does not result in the same degree of increase in bone mass as in postmenopausal osteoporosis.
Treatment/prevention of steroid-induced osteoporosis should begin with adequate calcium intake of 1000 to 1200 mg/day. Greater amounts are unlikely to be beneficial, and it probably is better to get the calcium from dietary sources instead of tablets. If the patient already has hypercalciuria, low-dose thiazides (12 mg/day) will reduce the hypercalciuria and allow more calcium intake.
The use of vitamin D in corticosteroid-treated osteoporosis is debated, and there is still not enough data to make strong recommendations. The most recent studies do not show a difference in bone density with active vitamin D metabolites or high doses of ergocalciferol. However, patients should receive some vitamin D to prevent deficiency (1000 units/day). In those cases with low urine calcium or clinical malabsorption or renal insufficiency the more potent vitamin D should be used (such as calcitriol at 0.25 mcg/day as a beginning dose).
Gonadal steroids (estrogen in women and testosterone in men) should be replaced as appropriate unless there are contraindications.
Anti-resorptive therapy can prevent some of the bone loss. Calcitonin nasal spray (one puff = 200 units/day) or bisphosphonates (I prefer alendronate 35mg/week) have been used. Clinical trials using bisphosphonates have shown benefits after one to two years; long-term studies are still needed. These therapies should not be used in patients with low serum calcium, and adequate dietary intake should be assured before they are started. Also avoid them in patients with reflux esophagitis, patients who are bedridden and can't be upright after taking the medication, and patients with renal insufficiency, and premenopausal women who may wish to become pregnant in the future.
The bone resorption may be high when steroids are initiated, but with longer use the osteoclasts are inhibited by the steroids so it does not make as much sense to continue anti-resorptive therapy. More recent studies comparing teriparatide to bisphosphonates have shown that there is significantly lower fracture rates in the patients treated with teriparatide. One of the studies lasted for 3 years and the P1NP was better than baseline throughout the study.
1. Teitelbaum SL, Seton MP, Saag KG. Should bisphosphonates be used for long-term treatment of glucocorticoid-induced osteoporosis? Arthritis and rheumatism. 2010. link
2. Teitelbaum SL. Glucocorticoids and the osteoclast. Clin Exp Rheumatol. 2015;33(4 Suppl 92):S37-9. link
3. Saag KG, Zanchetta JR, Devogelaer JP, Adler RA, Eastell R, See K, et al. Effects of teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis: thirty-six-month results of a randomized, double-blind, controlled trial. Arthritis and rheumatism. 2009;60(11):3346-55. link
4. Saag K, Shane E, Boonen S, Marin F, Donley DW, Taylor KA, et al. Teriparatide or Alendronate in Glucocorticoid-Induced Osteoporosis. The New England journal of medicine. 2007;357:2028-39. link
5. Pearce G, Tabensky DA, Delmas PD, Baker HW, Seeman E. Corticosteroid-induced bone loss in men. The Journal of clinical endocrinology and metabolism. 1998;83:801-6. link
6. Kim HJ, Zhao H, Kitaura H, Bhattacharyya S, Brewer JA, Muglia LJ, et al. Glucocorticoids suppress bone formation via the osteoclast. The Journal of clinical investigation. 2006;116(8):2152-60. link
7. Yao W, Dai W, Jiang L, Lay EY, Zhong Z, Ritchie RO, et al. Sclerostin-antibody treatment of glucocorticoid-induced osteoporosis maintained bone mass and strength. Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2015. link
8. Lems WF. Glucocorticoids: bad or safe for the bones? RMD open. 2015;1(Suppl 1):e000050. link
9. Hansen KE, Wilson HA, Zapalowski C, Fink HA, Minisola S, Adler RA. Uncertainties in the prevention and treatment of glucocorticoid-induced osteoporosis. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2011;26(9):1989-96. link
10. Adachi J, Saag K, Delmas P, Liberman U, Emkey R, Seeman E, et al. Two-Year Effects of Alendronate on Bone Mineral Density and Vertebral Fracture in Patients Receiving Glucocorticoids. Arthritis & Rheumatism. 2001;44:202-11. link
11. Yao W, Dai W, Jiang JX, Lane NE. Glucocorticoids and osteocyte autophagy. Bone. 2013;54(2):279-84. link
12. Van Staa TP, Laan RF, Barton IP, Cohen S, Reid DM, Cooper C. Bone density threshold and other predictors of vertebral fracture in patients receiving oral glucocorticoid therapy. Arthritis and rheumatism. 2003;48(11):3224-9. link
13. van Staa T, Leufkens HG, Abenhaim L, Zhang B, Cooper C. Oral corticosteroids and fracture risk: relationship to daily and cumulative doses. Rheumatology (Oxford, England). 2000;39:1383-9. link
14. Rizzoli R, Biver E. Glucocorticoid-induced osteoporosis: who to treat with what agent? Nature reviews Rheumatology. 2015;11(2):98-109. link
15. Piemontese M, Onal M, Xiong J, Wang Y, Almeida M, Thostenson JD, et al. Suppression of autophagy in osteocytes does not modify the adverse effects of glucocorticoids on cortical bone. Bone. 2015;75:18-26. link
16. Gluer CC, Marin F, Ringe JD, Hawkins F, Moricke R, Papaioannu N, et al. Comparative effects of teriparatide and risedronate in glucocorticoid-induced osteoporosis in men: 18-month results of the EuroGIOPs trial. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2013;28(6):1355-68. link
17. Cranney A, Welch V, Adachi JD, Homik J, Shea B, Suarez-Almazor ME, et al. Calcitonin for the treatment and prevention of corticosteroid-induced osteoporosis. Cochrane Database Syst Rev. 2000:CD001983. link
18. Bachrach BE. Bare-Bones Fact - Children are not small adults. The New England journal of medicine. 2004;351:924-6. link
19. van Staa T, Leufkens HG, Cooper C. The Epidemiology of Corticosteroid-Induced Osteoporosis: a Meta-analysis. Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2002;13:777-87. link
20. Shi J, Wang L, Zhang H, Jie Q, Li X, Shi Q, et al. Glucocorticoids: Dose-related effects on osteoclast formation and function via reactive oxygen species and autophagy. Bone. 2015;79:222-32. link
21. Sato H, Kondo N, Wada Y, Nakatsue T, Iguchi S, Fujisawa J, et al. The cumulative incidence of and risk factors for latent beaking in patients with autoimmune diseases taking long-term glucocorticoids and bisphosphonates. Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2015. link
22. Sato AY, Tu X, McAndrews KA, Plotkin LI, Bellido T. Prevention of glucocorticoid induced-apoptosis of osteoblasts and osteocytes by protecting against endoplasmic reticulum (ER) stress in vitro and in vivo in female mice. Bone. 2015;73:60-8. link
23. Manolagas SC. Steroids and osteoporosis: the quest for mechanisms. The Journal of clinical investigation. 2013;123(5):1919-21. link
24. Lodish MB, Hsiao H, Serbis A, Sinaii N, Rothenbuhler A, Keil MF, et al. Effects of Cushing Disease on Bone Mineral Density in a Pediatric Population. J Pediatr. 2010;156:1001-5. link
25. Leonard M, Feldman H, Shults J, Zemel B, Foster BJ, Stallings VA. Long-Term, High-Dose Glucocorticoids
and Bone Mineral Content in Childhood Glucocorticoid-Sensitive Nephrotic Syndrome. The New England journal of medicine. 2004;351:868-75. link
26. Lems WF, Saag K. Bisphosphonates and glucocorticoid-induced osteoporosis: cons. Endocrine. 2015;49(3):628-34. link
27. LeBlanc CM, Ma J, Taljaard M, Roth J, Scuccimarri R, Miettunen P, et al. Incident Vertebral Fractures and Risk Factors in the First Three Years Following Glucocorticoid Initiation Among Pediatric Patients With Rheumatic Disorders. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2015;30(9):1667-75. link
28. Kristo C, Jemtland R, Ueland T, Godang K, Bollerslev J. Restoration of the coupling process and normalization of bone mass following successful treatment of endogenous Cushing's syndrome: a prospective, long-term study. European journal of endocrinology / European Federation of Endocrine Societies. 2006;154(1):109-18. link
29. Kanis JA, Johansson H, Oden A, Johnell O, de Laet C, Melton IL, et al. A meta-analysis of prior corticosteroid use and fracture risk. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2004;19(6):893-9. link
30. Grossman JM, Gordon R, Ranganath VK, Deal C, Caplan L, Chen W, et al. American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis care & research. 2010;62(11):1515-26. link
31. Frenkel B. Glucocorticoid-induced osteoporosis. Advances in experimental medicine and biology. 2015;872:179-214. link
32. Feng Z, Zeng S, Wang Y, Zheng Z, Chen Z. Bisphosphonates for the prevention and treatment of osteoporosis in patients with rheumatic diseases: a systematic review and meta-analysis. PloS one. 2013;8(12):e80890. link
33. Eastell R, Chen P, Saag KG, Burshell AL, Wong M, Warner MR, et al. Bone formation markers in patients with glucocorticoid-induced osteoporosis treated with teriparatide or alendronate. Bone. 2010;46(4):929-34. link
34. Weinstein RS. Glucocorticoid-induced osteoporosis and osteonecrosis. Endocrinol Metab Clin North Am. 2012;41(3):595-611. link
35. Vestergaard P, Rejnmark L, Mosekilde L. Fracture risk associated with different types of oral corticosteroids and effect of termination of corticosteroids on the risk of fractures. Calcified tissue international. 2008;82(4):249-57. link
36. Van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C. Use of oral corticosteroids and risk of fractures. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2000;15(6):993-1000. link
37. Steinbuch M, Youket TE, Cohen S. Oral glucocorticoid use is associated with an increased risk of fracture. Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2004;15(4):323-8. link
38. Siu S, Haraoui B, Bissonnette R, Bessette L, Roubille C, Richer V, et al. Meta-analysis of tumor necrosis factor inhibitors and glucocorticoids on bone density in rheumatoid arthritis and ankylosing spondylitis trials. Arthritis care & research. 2015;67(6):754-64. link
39. Richy F, Ethgen O, Bruyere O, Reginster JY. Efficacy of alphacalcidol and calcitriol in primary and corticosteroid-induced osteoporosis: a meta-analysis of their effects on bone mineral density and fracture rate. Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2004;15(4):301-10. link
40. Kanis JA, Stevenson M, McCloskey EV, Davis S, Lloyd-Jones M. Glucocorticoid-induced osteoporosis: a systematic review and cost-utility analysis. Health technology assessment (Winchester, England). 2007;11(7):iii-iv, ix-xi, 1-231. link
41. Jacobs JW, Bijlsma JW, van Laar JM. Glucocorticoids in early rheumatoid arthritis: are the benefits of joint-sparing effects offset by the adverse effect of osteoporosis? the effects on bone in the utrecht study and the CAMERA-II study. Neuroimmunomodulation. 2015;22(1-2):66-71. link
42. Guanabens N, Gifre L, Peris P. The role of Wnt signaling and sclerostin in the pathogenesis of glucocorticoid-induced osteoporosis. Current osteoporosis reports. 2014;12(1):90-7. link
43. Dai Y, Hu S. Recent insights into the role of autophagy in the pathogenesis of rheumatoid arthritis. Rheumatology (Oxford, England). 2015. link
44. Dai W, Jiang L, Lay YA, Chen H, Jin G, Zhang H, et al. Prevention of glucocorticoid induced bone changes with beta-ecdysone. Bone. 2015;74:48-57. link
45. Ciccarelli F, De Martinis M, Ginaldi L. Glucocorticoids in patients with rheumatic diseases: friends or enemies of bone? Current medicinal chemistry. 2015;22(5):596-603. link
46. Briot K, Rouanet S, Schaeverbeke T, Etchepare F, Gaudin P, Perdriger A, et al. The effect of tocilizumab on bone mineral density, serum levels of Dickkopf-1 and bone remodeling markers in patients with rheumatoid arthritis. Joint, bone, spine : revue du rhumatisme. 2015;82(2):109-15. link
Therapy
References

