|
The bisphosphonates are powerful, they cause dramatic changes in the bone physiology, and they deserve respect. In women or men whose bone density T-score is lower than -2.5, or who already have a vertebral fracture, these medicines reduce the incidence of fractures and improve the quality of life. However, in women who don't have fractures and have only "osteopenia", a report by Schousboe found that alendronate is not cost-effective. We still don't know the effects of long-term suppression of bone formation.
| INDICATIONS |
|
Postmenopausal women with vertebral compression fractures Postmenopausal women with total hip bone density T-score below -2.5 Elderly men with non-traumatic fractures Some patients with secondary osteoporosis due to corticosteroids Paget's disease Cancer metastatic to bone Other bone diseases with high bone resorption |
| CONTRA-INDICATIONS |
Women who are pregnant or planning pregnancy Chronic kidney disease stages 4 or 5 Low serum calcium Osteomalacia Vitamin D deficiency (until it is corrected) Oral bisphosphonates should not be used in: Patients with serious esophageal disease Patients at bedrest who can't stay upright for an hour |
| USE WITH CAUTION |
Patients with high PTH Patients with gastric or intestinal bypass surgery Children (no long-term safety data) |
|
| |||||||||||||||||||||||||||||||||||||||||||||
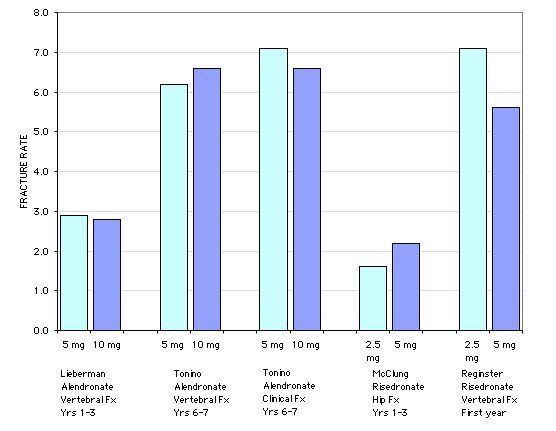
In the large trials the fracture rates with the lower doses were not significantly different from the rates in higher doses, despite greater increases in the DEXA measurements with the higher doses. In the risedronate study, with 5445 women between 70 and 79 yrs, the relative risk reduction for a hip fracture with the 2.5mg/d dose was statistically significant (CI 0.3 to 0.9) but for the 5mg/d dose it was not statistically signficant (CI 0.4 to 1.1). Nevertheless, the company decided to market the higher dose.
The FIT trial of alendronate documented significant fracture reduction at 2 years with the 5mg/day dose. After 2 years the dose was increased to 10mg/day, but the study design precluded actual dose comparisons.
Subsequent studies have shown that doses given once a week are as effective as those given daily. Almost all patients prefer this approach, so that is what I usually use.
Zoledronic acid continues to suppress bone formation and resorption for at least 2 years. A single dose increased the bone density at two years as well as two annual doses (McClung M).
| DOSE FOR OSTEOPOROSIS |
|---|
| Everybody needs adequate CALCIUM and VITAMIN D |
| Alendronate 35 mg once a week (this is the dose approved for prevention, and the official dose for treatment is 70mg a week) |
| Risedronate 15 mg a week more logical but approved dose is 35mg/week |
| Ibandronate 150mg/month oral OR 3mg/3 months I.V. push |
| Zoledronic acid: 4 or 5mg I.V. in 100ml NS over 30 minutes. A single dose lasts at least 2 years. Adjust for kidney function. Rapid infusion increases risk of renal damage. |
This is important!! These medications are poorly absorbed and any food in the stomach will reduce the amount aborbed. They also can cause esophageal erosions so must be "flushed down" with about 4 oz of water. In the FIT trial, a full glass of water caused more reflux and esophageal problems than 1/2 glass of water according to a lecture by Dr. David Karpf who was the clinical monitor for the study. Although it has not been carefully studied with newer medicines, calcium supplementation following the doses of etidronate reduced effectiveness, so I don't give any calcium for at least 4 hours after taking an oral bisphosphonate.
The Fracture Intervention Trial enrolled 6,000 postmenopausal women aged 55-79, with a bone density at the hip lower than 0.68g/cm2 (T-score about -2). There were two arms of the study: 2000 women who already had vertebral compression fractures and 4000 women who did not. The women who had a fracture took alendronate for three years, the others 4.2 years. Dose was 5mg/day for the first 2 years and then 10mg/day. The alendronate group showed increases in bone density of about 8% at the spine and 5% at the hip.
Summary of results of the Fracture Intervention Trial: Bone density increased in over 90% of women taking the medication, whether or not they already had a fracture. Notice how the incidence of new fractures depends on presence of baseline fracture. In women who did not have a vertebral fracture at baseline, the decrease in vertebral fractures was significant but of low magnitude, and the decrease in clinical fractures was not significant. In women who already had fractures (established osteoporosis) there was a decrease in fractures that was both statistically significant and clinically important.
The largest risedronate study included 9331 women: 5445 were 70-79 years old with T-score lower than -3 and 3886 were older than 80 with either low T-score or a clinical risk factor. BMD was done on 31% of the women older than 80. The graph shows the rates of hip fractures. Those with * were statistically significant.
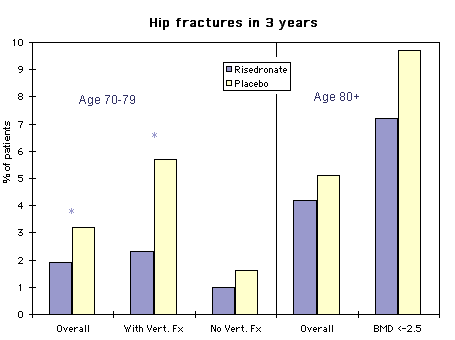
The incidence of hip fractures over time is shown in the graph which was modified from the one in the paper by putting both groups on the same scale. The older women had more fractures but no statistically significant benefit from the risedronate. 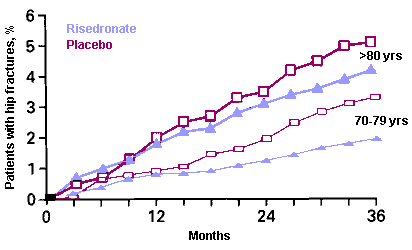
data from McClung, M. R.(2001). Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group. N Engl J Med 344: 333-40.
A nice review of the evidence from these large randomized trials was recently published by Bilezikian.
The FDA had a safety review of long-term use on Sept. 8, 2011. The extensive materials for this meeting are at this link .
I wrote an editorial review about this topic and you can download it from in the Cleveland Clinic Journal of Medicine.
The following graph shows change in bone density in 1099 women from the Fracture Intervention Trial who had taken alendronate for 5 years, and then were randomly given either alendronate or placebo for 5 years. The biochemical markers of bone resorption remained suppressed in both groups for 3 years after discontinuation. The serum formation markers were somewhat different; P1NP increased moderately but was still 24% below baseline 5 years after stopping, whereas the BAP gradually increased towards baseline.
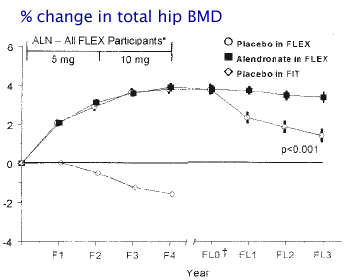 | 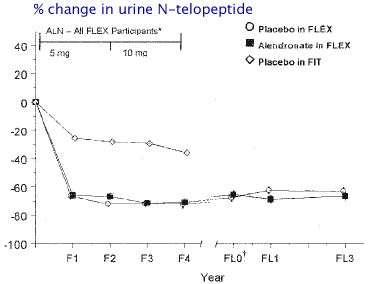 |
from Ensrud KE, Barrett-Connor EL, Schwartz A, et al. Randomized trial of effect of alendronate continuation versus discontinuation in women with low BMD: results from the Fracture Intervention Trial long-term extension. J Bone Miner Res. 2004;19(8):1259-69. Used with permission from American Society for Bone and Mineral Research.
Despite the difference in bone density during the last 5 years, the women who discontinued alendronate had essentially the same number of fractures as the women who kept taking the drug (Black DM). The alendronate group had fewer "clinical" vertebral fractures (those diagnosed by their physician) but when the xrays were measured the total number of "morphometric" fractures was not different. Also, there was no difference in moderate to severe vertebral fractures. Among the women with fractures there was more height loss in the alendronate group (3.5cm) vs the placebo group (2.1cm, p=.02)(Ott SM).
Because there is no benefit to reduce fractures beyond 5 years, it is logical to stop alendronate after 5 years in most patients. Currently there are no practice guidelines about this issue and experts disagree about when to stop bisphosphonates.
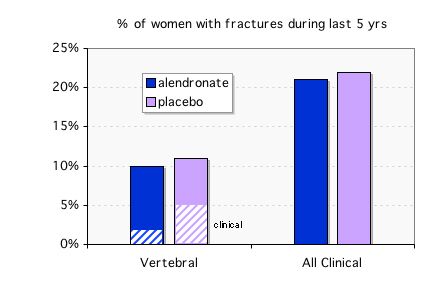
 A post-hoc sub-group analysis suggested a benefit to staying on alendronate in women with osteoporosis who have not had a fracture, but the data is not very robust. Click to read even more details about the FLEX study and how people slightly mis-report the data in a way that is favorable to alendronate.
A post-hoc sub-group analysis suggested a benefit to staying on alendronate in women with osteoporosis who have not had a fracture, but the data is not very robust. Click to read even more details about the FLEX study and how people slightly mis-report the data in a way that is favorable to alendronate.
Meijer used data from the PHARMO database in the Netherlands to examine the fracture rates in all 14,750 women who started a bisphoshonate for osteoporosis between 1996 and 2004. Over half the women stopped the bisphosphonate during the first year. There were 387 cases who were hospitalized for a fracture. Those who took bisphosphonates for 3 to 4 years had significantly fewer fractures than those who stopped during the first year (Odds ratio 0.54). However, those who took bisphosphonates for 5 to 6 years had slightly more fractures than those who took them for less than a year.
Click  here to see another graph from a ten-year study by
H. Bone. The fracture rate in those who remained on the alendronate for ten years was about the same during the later years as in the first three years, so there did not seem to be any evidence of harm for up to ten years of use. There were no controls after year 3 so it is not possible to be sure about benefits or risks beyond 3 years.
here to see another graph from a ten-year study by
H. Bone. The fracture rate in those who remained on the alendronate for ten years was about the same during the later years as in the first three years, so there did not seem to be any evidence of harm for up to ten years of use. There were no controls after year 3 so it is not possible to be sure about benefits or risks beyond 3 years.
We don't know because the alendronate was first on the market in 1996 and there are not many patients who have taken the medications longer than ten years.
Bone biopsies from patients at ten years show very low bone formation rates. In the FLEX study the mineralizing surface was 1.4 +/- 1.6% in 9 samples. All had some double labels, but they were very short. Another study in women treated with bisphosphonates longer than 4 years found 33% of patients had no double labels ( Chapurlat RD). No increase in micro-cracks were seen in these women, however. In dogs, micro-cracks are seen even when doses are similar to those in humans, and another study of women on long-term bisphosphoantes did see evidence of increased cracks (Dobnig.) At high doses there are negative effects on the toughness of the bone, but at lower doses there are not measureable biomechanical defects. (Allen MR). Also, the micro-cracks in beagles do not continue to accumulate after the first year (Allen, ASBMR 06).
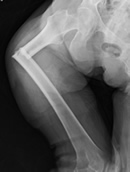
| So far, there is no convincing direct evidence that bisphosphonates cause bone problems after ten years in humans (Liberman UA). Several recent reports, however, suggest that subtrochanteric fractures appear to be related to oversuppression of bone formation associated with bisphosphonates. These are unusual fractures. Click on the image for more details. |
The clinical trials of bisphosphonates have not reported any increased incidence of fracture non-union in patients treated with active drug. However, Solomon recently found an association between non-union of humerus fractures and bisphoshonates.
When bisphosphonates are given to patients after joint replacement surgery, there is less loosening of the prosthesis (Arabmotlagh M, Hilding M, Wilkinson JM) although after 5 years there was no residual positive effect of a dose of pamidronate given at the time of surgery (Shetty N). When given to patients 2 weeks after a fracture of the lower leg, bisphosphonates prevented the bone loss that was seen in the proximal femur of placebo control patients (van der Poest Clement).
Animal studies show that callus forms vigourously in animals who had been given bisphosphonates prior to the fracture (Peter CP, Cao Y, Li C). However, the callus does not normally remodel. The bisphosphonate-treated fracture callus persists, and woven bone does not get replaced by lamellar bone. When alendronate was given to pigs after spine fusion with a bone graft, the fusion area had more woven bone and greater amount of fibrous tissue, with no difference in the rate of fusion (Xue Q) Another study in rats, however, found that despite the higher fusion area, the fusion rate was lower in alendronate-treated groups (Huang RC).
After a fragility fracture (for example, a hip fracture) in an untreated patient with osteoporosis, it makes sense to begin a bisphosphonate. The demonstrated risk of a future fracture is greater than the potential risk of non-union or poor callus remodelling. Of course, these patients need an evaluation for other causes, and concomitant treatment with calcium and vitamin D and physical therapy. It is possible that treatment with anabolic agents will provide even better benefit for the skeleton, but currently bisphosphonates remain the first choice due to their lower cost and greater familiarity.
In the recent study of zoledronic acid following a hip fracture, Eriksen EF found that patients dosed later than 6 wk after hip fracture exhibited a greater increase in total hip and femoral neck BMD at month 12 compared with patients dosed earlier than 6 wk. The subjects who were treated immediately after the surgery did not respond as well, although it is possible that they were more frail. An animal study by Amanat N found better fracture healing when zoledronic acid treatment was delayed.
 Click to see larger image. |
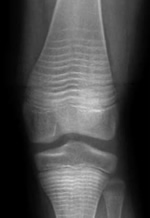
Children with severe osteogenesis imperfecta, who have multiple fractures, show reduction in pain and fracture rates with bisphosphonates. The radiographs of the long bones show a unique striped pattern when pamidronate is given intermittently, and this is caused by layers of thick bone alternating with osteopenic bone. There may be some weakness in these areas. Currently it is unclear when to stop giving these medications. The drugs are still excreted in the urine 8 years after stopping. Because of uncertainties about long-term effects, these drugs should be used only in serious cases. from Land C, Rauch F, Glorieux FH. Cyclical intravenous pamidronate treatment affects metaphyseal modeling in growing patients with osteogenesis imperfecta. J Bone Miner Res. 2006;21(3):374-9.
Used with permission from American Society for Bone and Mineral Research
Children with polyostotic fibrous dysplasia or juvinile Paget's disease may also benefit from bisphosphonates. Again, there are uncertainties about how long to use the medications (Speiser PW). |
Bisphosphonates are NOT APPROVED for prevention of osteoporosis in premenopausal women. They should not be used in women who got a DEXA out of curiosity and discovered osteopenia. They are beneficial in other situations, such as prolonged high dose steroid use, organ transplantation, fibrous dysplasia, and metastatic carcinoma. Studies in animals show fetal and maternal abnormalities in bones and calcium metabolism, so it is unethical to study this medication in pregnant women or women who might become pregnant while the bisphosphonate is still in the bones.
The following graph shows the effect on bone density in women who are recently postmenopausal. Alendronate 5mg/day increases bone density compared to placebo, but not as well as estrogen with norethindrone or estrogen with medroxyprogesterone. These results are from data in the EPIC study (Ravn). There were very few fractures in this study, and no significant difference in fracture rates could be detected.
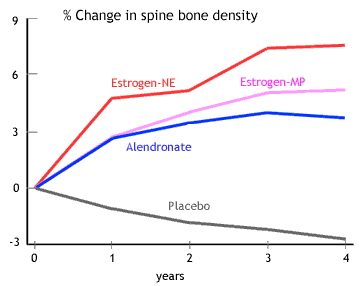 | Click on the graph to see what happened when women took alendronate for 2 years and then stopped (shown in the dashed grey line). |
Many experts say that bisphosphonates could be used instead of estrogen in women with osteopenia, to prevent osteoporotic fractures. This is based on wishful thinking instead of evidence. It takes decades to reach "the age of fracture" and we don't know if any drugs except estrogen will work that long. In my opinion, bisphosphonates should be used only if the risk of fracture within the next ten years is high enough to justify the potential risks. As more evidence accumulates about long-term benefits, my recommendations may change. I also wonder if very small doses might be better for long-term prevention, but I'm not aware of any ongoing studies.
In the Fracture Intervention Trial the women between 75 and 80 who had established osteoporosis showed improvement in bone density and fracture reduction that was similar to the women aged 55-75. One study of alendronate included women until age 85, but mean age was 71 so most women were still younger than 80. In that study there were not enough women to determine fracture rates, but the bone density increased 6% at the spine and 4% at the femoral trochanter.
Risedronate studies shown above did not show fracture benefit to women older than 80, even among the 941 women who had osteoporosis by bone densitometry. Maybe this was just bad luck, with a negative finding due to inadequate power. Perhaps in the older women, other factors (such as quality of bones or falls) become relatively more important so it is more difficult to determine a beneficial drug effect. Some women may have lost so much bone mass by age 80 that they will have fractures despite improvement in the bone strength. There is also a possibility that the bisphosphonate does not work as well in women older than 80, because these women may already have low bone formation rate due to inadequate osteoblasts.
In the recent report of zoledronate the average age was 73 and women up to age 89 were enrolled. These women with established osteoporosis had a significant reduction in hip fractures.
Bisphosphonates are excreted by the kidneys and are not recommended for persons with stage 4 or 5 chronic kidney disease. Often elderly, thin women with low muscle mass have a deceptively normal creatinine. This should be taken into account when prescribing bisphosphonates. Also, it is especially important to insure adequate vitamin D levels in elderly women before starting bisphosphonates, because the skin is less effective at converting vitamin D after sunlight exposure.
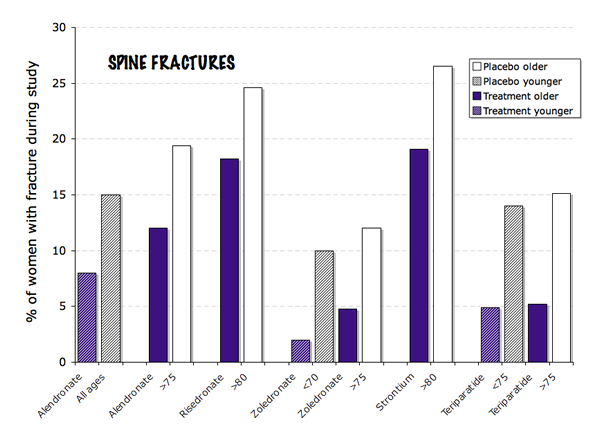 This image shows the improvement in rate of spine fractures with various medicines in elderly subjects. Some of the trials compared young to older women, and with alendronate and zoledronate the younger women had a better response.
This image shows the improvement in rate of spine fractures with various medicines in elderly subjects. Some of the trials compared young to older women, and with alendronate and zoledronate the younger women had a better response.
|
Bisphosphonate structures and bone effects |  |
Updated 7/3/2013

