Bisphosphonate structure

The bisphosphonates are all related to pyrophosphate (PPi), which is a byproduct of cellular metabolism.
When nucleotides are formed, then:
ATP -> AMP + PPi
Pyrophosphate is a natural circulating inhibtor of mineralization in the blood and urine which can't get inside the bones because the lining cells destroy it with alkaline phosphatase. People with genetic problems of alkalline phosphatase have osteomalacia, because the pyrophoshate can get into the bone and it prevents mineralization.
If a carbon is substituted for the oxygen, then a bisphosphonate is formed. The bisphosphonates not only get inside the bone, but they attach very strongly to the bone mineral. The different strengths of the bisphosphonates depend on the variations in the side chains. Notice how similar some of these drugs are to each other.
Click  to see a more formal image of bisphosphonate structures. to see a more formal image of bisphosphonate structures.
Anti-resorptive potency
of different bisphosphonates
| | Drug |
Potency
|
|---|
| etidronate | 1
|
|---|
| clodronate | 10
|
|---|
| tiludronate | 10
|
|---|
| pamidronate | 100
|
|---|
| alendronate | 1000
|
|---|
| risedronate | 5000
|
|---|
| ibandronate | 10000
|
|---|
|
Physiological effects
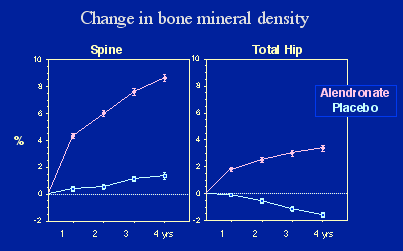
| The bisphosphonates increase bone mass in both cortical and trabecular bone. In this slide from the FIT study, the dose was doubled at year 2.
|
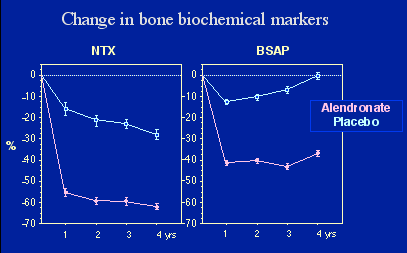
| The biochemical markers of both bone resorption (N-telopeptide) and bone formation (Bone specific alkaline phosphatase) show decreases with the bisphosphonates.
|
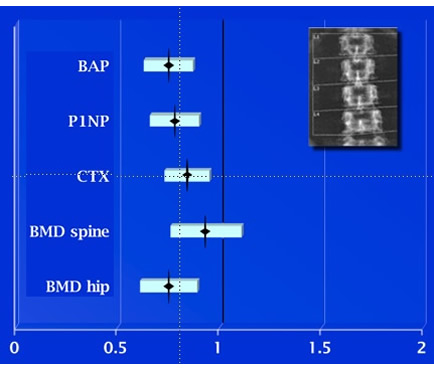
| This study by Bauer shows that in women treated with alendronate the change in biochemical markers predicts the incidence of spine fractures as well or better than the change in bone density. Shown is the relative risk of a fracture for each standard deviation decrease in the markers or increase in the DEXA values.
 Move your mouse over the image to see the results for hip fractures, in which only the bone alkaline phosphatase change predicted fracture outcome. Move your mouse over the image to see the results for hip fractures, in which only the bone alkaline phosphatase change predicted fracture outcome.
|
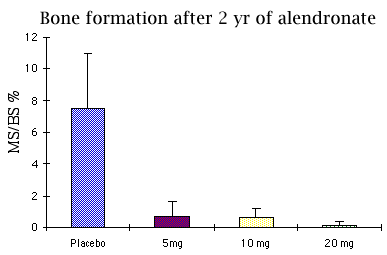
| The bisphosphonates cause dramatic reductions in the bone formation. This is seen on bone biopsies labelled with tetracycline. It is not clear if this is a direct effect of bisphosphonates or if it is just secondary to the inhibition of bone resorption. The graph shows the mineralizing surface as a percentage of the trabecular bone surface. With higher doses only 0.16% of the surface is still forming bone (Chavassieux). Below is seen a graph of published studies that report mineralization surfaces in normals and in patients treated with bisphosphonates. |

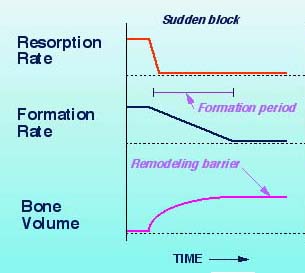 | EFFECTS OF BLOCKING RESORPTION
Bisphosphonates block resorption, but formation continues for a forming period (6-12 months). During that interval the bone volume (and bone mass) will increase. This is a one-time increase, and afterwards the bone volume will plateau.
The slight increases seen after a year probably represent increased mineralization within each newly formed bone remodeling unit. The mineral is more densely packed and so the bone density will increase even though the bone volume does not. The major effect is to increase bone strength, and this is seen in clinical trials up to five years duration. After prolonged usage or with high doses the bone could potentially become more brittle.
|
Overall, the major way that bisphosphonates improve bone strength is that they prevent trabecular perforations that lead to weakened micro-structure. While the bone volume may increase a small amount, most studies of human bone biopsies have not been able to demonstrate this. The following diagram shows a summary of the bisphosphonate actions, with question marks showing mechanisms that are still uncertain. More information about bone perforations is on the page about bone quality.

The bisphosphonate effects are shown graphically in these

Animations
Cellular mechanisms of action
Etidronate may act by causing chemical changes to the mineral itself, inhibiting crystal formation and making the mineral resistant to resorption. These effects depend on the amount of drug given. This is why etidronate can be used to treat heterotopic calcifications, or prevent vascular calcifications in animal models. The trouble is that prolonged use results in osteomalacia. These "first generation" bisphosphonates can inhbit the ATP. With the newer bisphosphonates, the dose is so small that the effects cannot be caused by physical-chemical changes in the mineral, and osteomalacia is a rare side effect.
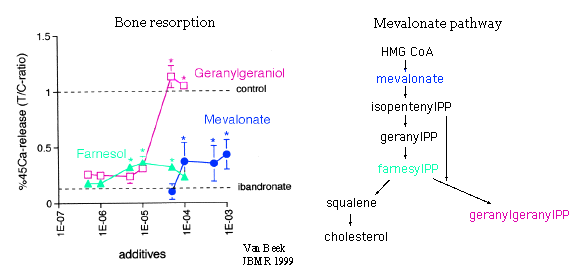
These figures from van Beek show the mevalonate pathway of cholesterol synthesis. HMG CoA reductase inhibitors such as mevastatin, used to treat hypercholesterolemia, had been shown to reduce bone resorption. Ibandronate inhibited bone resorption (shown as the lower dotted line). When geranylgeraniol was added, the inhibition was reversed. This was seen in the nitrogen-containing bisphosphonates (risedronate, alendronate, olpadronate, & ibandronate). GeranylgeranylPP modifies (prenylates) a small GTP-binding protein called rho21, which regulates the cytoskeleton. Thus, low levels of geranlygeranylPP lead to abnormalies in the cytoskeleton, so osteoclasts don't have a ruffled border and are unable to resorb bone.
The nitrogen-containing bisphosphonates inhibit the farnesyl pyrophosphate synthase.
A beautiful review of the actions, showing how the drug fits into the enzymatic site of the proteins, and how the angles determine the tightness of binding to the mineral, is by Russell RG.
Updated 3/26/10



 to see a more formal image of bisphosphonate structures.
to see a more formal image of bisphosphonate structures.



 Move your mouse over the image to see the results for hip fractures, in which only the bone alkaline phosphatase change predicted fracture outcome.
Move your mouse over the image to see the results for hip fractures, in which only the bone alkaline phosphatase change predicted fracture outcome.







