Bone Histomorphometry
Summary of bone biopsy findings
|
The teriparatide effects are shown graphically in these
Animations |
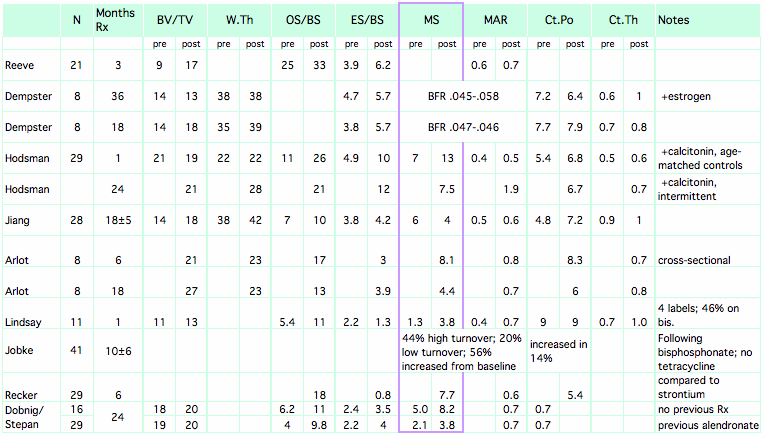 This table shows results before and after teriparatide, or in controls vs. treated patients.
This table shows results before and after teriparatide, or in controls vs. treated patients. Columns are blank for the studies that did biopsies only after treatment.
Physiological actions
The role of PTH in control of bone mass is still not really understood. PTH stimulates osteoblastic activity, especially on trabecular surfaces. It also stimulates osteoclastic activity. In some cases the anabolic effect predominates over the increased resorption, and osteosclerosis results. Intermittent spikes of PTH, such as given by daily injection, will cause more increase in bone formation than in bone resorption. This has been shown in rats. Furthermore, micro-array analysis has shown that different genes are expressed in bone cells that have been exposed to continuous PTH than those expressed after intermittent PTH (Onyia JE).
PTH has different effects on the cortical and trabecular bone. Patients with either primary or secondary hyperparathyroidism have increased bone density of the spine, but decreased cortical bone mass. Patients with osteoporosis treated with PTH showed increases at the spine but decreases at cortical sites, especially the radius. Total body calcium decreased about 2%. QCT images demonstrated increases in cancellous bone of the hip, and overall increased amount of cortical bone, but the cortical bone became more porous.
The anabolic actions of PTH diminish with time; after a mean of 19 months, the bone forming surface from bone biopsies is slightly lower than at baseline. Studies using 85-Strontium kinetics, however, suggest that other parts of the skeleton might still be forming bone even when the iliac crest has returned to baseline (Reeve).
Intermittent PTH suppresses apoptosis of the osteoblasts (Jilka) which is one reason there is a high bone formation rate. On the other hand, continuous PTH will increase the apoptosis (Turner). The mechanisms of the anabolic effect are not really certain; it appears that intermittent PTH can inhibit SOST (Keller H). Also, osteoblast PTHrP influences the action of PTH 1-34; in mice with heterozygous deficiency of PTHrP in osteoblasts, the anabolic effect of PTH is enhanced compared to wild-type mice (Miao D).
An interesting report by Shao JS demonstrated decreased vascular calcifications in genetically predisposed mice, after treatment with teriparatide. The mechanism was an inhibition in the transcription factor Msx, which resulted in decreased Wnt signalling. This suggests that intermittent PTH might have different effects on Wnt signalling, depending on the cell type.
Chavassieux P reported a case of a patient with pycnodysostosis who was treated with teriparatide. There was no effect. This disease is caused by a mutation in the gene for cathepsin K, so that osteoclasts are unable to resorb the bone. The authors suggest that some aspects of osteoclast function are necessary for the anabolic effect of teriparatide.
A recent biopsy study in persons who took teriparatide found that microcrack damage was significantly improved in the patients who had previously been taking alendronate (Dobnig H).
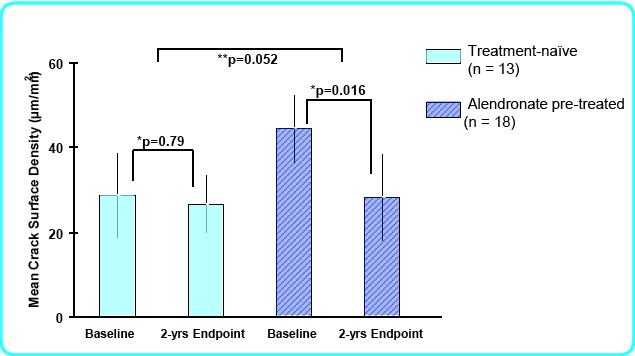 in Dobnig H, Stepan JJ, Burr DB, et al. Teriparatide Reduces Bone Microdamage Accumulation in Postmenopausal Women Previously Treated with Alendronate. J Bone Miner Res 2009, online May 19. Used with permission from ASBMR
in Dobnig H, Stepan JJ, Burr DB, et al. Teriparatide Reduces Bone Microdamage Accumulation in Postmenopausal Women Previously Treated with Alendronate. J Bone Miner Res 2009, online May 19. Used with permission from ASBMR
Reviews about PTH:
Hodsman AB. Parathyroid hormone and teriparatide for the treatment of osteoporosis: a review of the evidence and suggested guidelines for its use. Endocr Rev 2005;26(5):688-703.
Poole KE. Parathyroid hormone - a bone anabolic and catabolic agent. Curr Opin Pharmacol 2005;5(6):612-7.

